Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Sato, Tetsuya; Nagame, Yuichiro*
Nihon Butsuri Gakkai-Shi, 78(2), p.64 - 72, 2023/02
The study of the chemistry of superheavy elements, which are located in the heavy extremes of the periodic table, has made considerable progress over the past 20 years, and new approaches based on various ideas have recently been developed. Research groups in Japan have also made significant contributions to the development of research on superheavy elements. Recently, notable results have been reported for the transactinide elements rutherfordium (element 104), dubnium (element 105), and seaborgium (element 106), and the heavy actinides with atomic numbers exceeding 100. The review will focus on the recent main results of these elements. This review outlines the main recent results and touches on future prospects.
G tz, M.*; Yakushev, A.*; G
tz, M.*; Yakushev, A.*; G tz, S.*; Di Nitto, A.*; D
tz, S.*; Di Nitto, A.*; D llmann, Ch. E.*; Asai, Masato; Kindler, B.*; Krier, J.*; Lommel, B.*; Nagame, Yuichiro*; et al.
llmann, Ch. E.*; Asai, Masato; Kindler, B.*; Krier, J.*; Lommel, B.*; Nagame, Yuichiro*; et al.
Radiochimica Acta, 110(2), p.75 - 86, 2022/02
Times Cited Count:2 Percentile:31.78(Chemistry, Inorganic & Nuclear)The study of volatile superheavy element carbonyl complexes requires more efficient methods because the yield of transactinide elements decreases with increasing atomic number. This is achieved by using a newly developed double chamber system to separate the recoil chamber and the reaction one, thereby avoiding the decomposition of reactive molecules by the projectile ion beam, which hinders the synthesis of carbonyl complexes. The feasibility of this method was verified by synthesizing 5d metal short-lived isotopes as homologous element isotopes of the light transactinide elements Sg, Bh, Hs, and Mt at the Japan Atomic Energy Agency tandem accelerator and conducting model experiments.
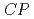 -violation studies on superheavy molecules; Theoretical and experimental perspectives
-violation studies on superheavy molecules; Theoretical and experimental perspectivesMitra, R.*; Prasannaa, V. S.*; Garcia Ruiz, R. F.*; Sato, Tetsuya; Abe, Minori*; Sakemi, Yasuhiro*; Das, B. P.*; Sahoo, B. K.*
Physical Review A, 104(6), p.062801_1 - 062801_9, 2021/12
Times Cited Count:2 Percentile:24.82(Optics)We provide detailed theoretical studies of quantities relevant to the electron electric dipole moment (eEDM) and nucleus-electron scalar-pseudoscalar interactions in diatomic molecules containing superheavy lawrencium nuclei. The sensitivity to parity and time (or, equivalently, 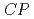 ) reversal violating properties is studied for different neutral and ionic molecules. The effective electric fields in these systems are found to be about 3
) reversal violating properties is studied for different neutral and ionic molecules. The effective electric fields in these systems are found to be about 3 4 times larger than other known molecules on which eEDM experiments are being performed. Similarly, these superheavy molecules exhibit an enhancement of more than 3 times for
4 times larger than other known molecules on which eEDM experiments are being performed. Similarly, these superheavy molecules exhibit an enhancement of more than 3 times for 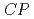 -violating scalar-pseudoscalar nucleus-electron interactions. Our preliminary analysis using the Woods-Saxon nuclear model also demonstrates that these results are sensitive to the diffuse surface interactions inside the Lr nucleus.
-violating scalar-pseudoscalar nucleus-electron interactions. Our preliminary analysis using the Woods-Saxon nuclear model also demonstrates that these results are sensitive to the diffuse surface interactions inside the Lr nucleus.
Yakushev, A.*; Lens, L.*; D llmann, C. E.*; Block, M.*; Nagame, Yuichiro*; Sato, Tetsuya; Toyoshima, Atsushi*; 42 of others*
llmann, C. E.*; Block, M.*; Nagame, Yuichiro*; Sato, Tetsuya; Toyoshima, Atsushi*; 42 of others*
Frontiers in Chemistry (Internet), 9, p.753738_1 - 753738_9, 2021/11
Times Cited Count:10 Percentile:63.67(Chemistry, Multidisciplinary)Nihonium (Nh, element 113) and flerovium (Fl, element 114) are the first superheavy elements in which the 7p shell is occupied. High volatility and inertness were predicted for Fl due to the strong relativistic stabilization of the closed 7p sub-shell, which originates from a large spin-orbit splitting between the 7p
sub-shell, which originates from a large spin-orbit splitting between the 7p and 7p
and 7p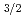 orbitals. One unpaired electron in the outermost 7p
orbitals. One unpaired electron in the outermost 7p sub-shell in Nh is expected to give rise to a higher chemical reactivity. Theoretical predictions of Nh reactivity are discussed, along with results of the first experimental attempts to study Nh chemistry in the gas phase. The experimental observations verify a higher chemical reactivity of Nh atoms compared to its neighbor Fl and call for the development of advanced setups. First tests of a newly developed detection device miniCOMPACT with highly reactive Fr isotopes assure that effective chemical studies of Nh are within reach.
sub-shell in Nh is expected to give rise to a higher chemical reactivity. Theoretical predictions of Nh reactivity are discussed, along with results of the first experimental attempts to study Nh chemistry in the gas phase. The experimental observations verify a higher chemical reactivity of Nh atoms compared to its neighbor Fl and call for the development of advanced setups. First tests of a newly developed detection device miniCOMPACT with highly reactive Fr isotopes assure that effective chemical studies of Nh are within reach.

Chiera, N. M.*; Sato, Tetsuya; Eichler, R.*; Tomitsuka, Tomohiro; Asai, Masato; Adachi, Sadia*; Dressler, R.*; Hirose, Kentaro; Inoue, Hiroki*; Ito, Yuta; et al.
Angewandte Chemie; International Edition, 60(33), p.17871 - 17874, 2021/08
Times Cited Count:2 Percentile:14.88(Chemistry, Multidisciplinary)The formation and the chemical characterization of single atoms of dubnium (Db, element 105), in the form of its volatile oxychloride, was investigated using the on-line gas phase chromatography technique, in the temperature range 350 - 600  C. Under the exact same chemical conditions, comparative studies with the lighter homologs of group-5 in the Periodic Table clearly indicate the volatility sequence being NbOCl
C. Under the exact same chemical conditions, comparative studies with the lighter homologs of group-5 in the Periodic Table clearly indicate the volatility sequence being NbOCl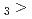 TaOCl
TaOCl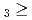 DbOCl
DbOCl . From the obtained experimental results, thermochemical data for DbOCl
. From the obtained experimental results, thermochemical data for DbOCl were derived. The present study delivers reliable experimental information for theoretical calculations on the chemical properties of transactinides.
were derived. The present study delivers reliable experimental information for theoretical calculations on the chemical properties of transactinides.
G tz, M.*; G
tz, M.*; G tz, S.*; Kratz, J.-V.*; Ballof, J.*; D
tz, S.*; Kratz, J.-V.*; Ballof, J.*; D llmann, Ch. E.*; Eberhardt, K.*; Mokry, C.*; Renisch, D.*; Runke, J.*; Sato, Tetsuya; et al.
llmann, Ch. E.*; Eberhardt, K.*; Mokry, C.*; Renisch, D.*; Runke, J.*; Sato, Tetsuya; et al.
Radiochimica Acta, 109(3), p.153 - 165, 2021/03
Times Cited Count:3 Percentile:45.99(Chemistry, Inorganic & Nuclear)We report on a novel two-chamber approach for the synthesis of volatile complexes that allows spatial decoupling of thermalization and gas-phase carbonyl complex synthesis. Neutron induced fission on  U and spontaneous fission of
U and spontaneous fission of  Cm were employed for the production of the fission products. These were stopped inside a gas volume behind the target and flushed with an inert-gas flow into a second chamber. This was flushed with carbon monoxide to allow the gas-phase synthesis of carbonyl complexes. Parameter studies of the transfer from the first into the second chamber as well as on the carbonyl complex formation and transport processes have been performed. High overall efficiencies of more than 50% were reached rendering this approach interesting for studies of superheavy elements. Our results show that carbonyl complex formation of thermalized fission products is a single-atom reaction, and not a hot-atom reaction.
Cm were employed for the production of the fission products. These were stopped inside a gas volume behind the target and flushed with an inert-gas flow into a second chamber. This was flushed with carbon monoxide to allow the gas-phase synthesis of carbonyl complexes. Parameter studies of the transfer from the first into the second chamber as well as on the carbonyl complex formation and transport processes have been performed. High overall efficiencies of more than 50% were reached rendering this approach interesting for studies of superheavy elements. Our results show that carbonyl complex formation of thermalized fission products is a single-atom reaction, and not a hot-atom reaction.
 Bh in the
Bh in the  Cm(
Cm(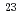 Na,5
Na,5 )
) Bh reaction and its decay properties
Bh reaction and its decay propertiesHaba, Hiromitsu*; Fan, F.*; Kaji, Daiya*; Kasamatsu, Yoshitaka*; Kikunaga, Hidetoshi*; Komori, Yukiko*; Kondo, Narumi*; Kudo, Hisaaki*; Morimoto, Koji*; Morita, Kosuke*; et al.
Physical Review C, 102(2), p.024625_1 - 024625_12, 2020/08
Times Cited Count:6 Percentile:59.56(Physics, Nuclear)Tanaka, Taiki*; Morita, Kosuke*; Morimoto, Koji*; Kaji, Daiya*; Haba, Hiromitsu*; Boll, R. A.*; Brewer, N. T.*; Van Cleve, S.*; Dean, D. J.*; Ishizawa, Satoshi*; et al.
Physical Review Letters, 124(5), p.052502_1 - 052502_6, 2020/02
Times Cited Count:19 Percentile:81.08(Physics, Multidisciplinary)Sato, Tetsuya
Kagaku To Kogyo, 72(10), P. 867, 2019/10
We conducted measurements of the first ionization potential (IP ) of the heavy actinide elements, lawrencium (Lr,
) of the heavy actinide elements, lawrencium (Lr, 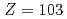 ), nobelium (No,
), nobelium (No, 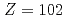 ), mendelevium (Md,
), mendelevium (Md, 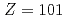 ) and fermium (Fm,
) and fermium (Fm, 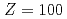 ) by using a novel method based on a surface ionization process. The IP
) by using a novel method based on a surface ionization process. The IP measurements have been performed using the ISOL (Isotope Separator On-Line) system equipped with a surface ion-source with short-lived heavy actinide isotopes,
measurements have been performed using the ISOL (Isotope Separator On-Line) system equipped with a surface ion-source with short-lived heavy actinide isotopes,  Lr (
Lr (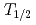 = 27s),
= 27s), 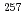 No (
No (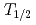 = 24.5s),
= 24.5s), 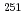 Md (
Md (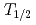 = 4.27 min), and
= 4.27 min), and  Fm (
Fm (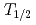 = 2.6 min). Our experimental results clearly showed that the IP
= 2.6 min). Our experimental results clearly showed that the IP of Lr is distinctly low among actinide elements. Moreover, No has the highest IP
of Lr is distinctly low among actinide elements. Moreover, No has the highest IP among them due to its full-filled 5f and 7s orbitals; the IP
among them due to its full-filled 5f and 7s orbitals; the IP value increased with an atomic number up to No and decreased dramatically at Lr, indicating the similar trend with that of heavy lanthanide elements. Therefore, we concluded Lr would be the last member of the actinide series.
value increased with an atomic number up to No and decreased dramatically at Lr, indicating the similar trend with that of heavy lanthanide elements. Therefore, we concluded Lr would be the last member of the actinide series.
Chiera, N. M.; Sato, Tetsuya; Tomitsuka, Tomohiro; Asai, Masato; Ito, Yuta; Shirai, Kaori*; Suzuki, Hayato; Tokoi, Katsuyuki; Toyoshima, Atsushi; Tsukada, Kazuaki; et al.
Journal of Radioanalytical and Nuclear Chemistry, 320(3), p.633 - 642, 2019/06
Times Cited Count:1 Percentile:11.15(Chemistry, Analytical)An isothermal gas-chromatographic (IGC) device has been developed and tested for on-line gas phase studies of volatile oxychlorides of short-lived group-5 transition metals. Radioisotopes of niobium and tantalum, produced in nuclear fusion evaporation reactions, are directly flushed into the IGC setup by an inert gas-jet. Oxychloride compounds are formed by the addition of SOCl and O
and O . Parameters influencing the formation and transport of NbOCl
. Parameters influencing the formation and transport of NbOCl and TaOCl
and TaOCl are investigated. For nuclides with half-lives (
are investigated. For nuclides with half-lives (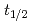 ) of about 30 s, an overall efficiency of 7% is obtained, rendering the IGC setup suitable for the chemical exploration of
) of about 30 s, an overall efficiency of 7% is obtained, rendering the IGC setup suitable for the chemical exploration of  Db(
Db(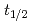 = 34s).
= 34s).
Chiera, N. M.; Sato, Tetsuya; Tomitsuka, Tomohiro; Asai, Masato; Suzuki, Hayato*; Tokoi, Katsuyuki; Toyoshima, Atsushi; Tsukada, Kazuaki; Nagame, Yuichiro
Inorganica Chimica Acta, 486, p.361 - 366, 2019/02
Times Cited Count:3 Percentile:18.6(Chemistry, Inorganic & Nuclear)The formation of NbOCl and TaOCl
and TaOCl and their adsorption behavior on quartz surfaces was explored by applying an isothermal gas-chromatographic method. Trace amounts of short-lived Nb and Ta isotopes were used. Adsorption enthalpy values (
and their adsorption behavior on quartz surfaces was explored by applying an isothermal gas-chromatographic method. Trace amounts of short-lived Nb and Ta isotopes were used. Adsorption enthalpy values ( ) at zero surface coverage of -
) at zero surface coverage of - (NbOCl
(NbOCl ) = 102
) = 102  4 kJ/mol and -
4 kJ/mol and - (TaOCl
(TaOCl ) = 128
) = 128  5 kJ/mol were determined by analyzing the chromatographic behavior of the Nb andTa complexes with a Monte-Carlo simulation method based on an adsorption-desorption kinetic model.By applying an empirical correlation, the experimental
5 kJ/mol were determined by analyzing the chromatographic behavior of the Nb andTa complexes with a Monte-Carlo simulation method based on an adsorption-desorption kinetic model.By applying an empirical correlation, the experimental  values were successively related to the macroscopic standard sublimation enthalpy,
values were successively related to the macroscopic standard sublimation enthalpy, 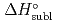 , as a measure of the volatility of each substance. The inferred sublimation enthalpies are in agreement with tabulated thermochemical values. Thus, the linear empirical correlation between
, as a measure of the volatility of each substance. The inferred sublimation enthalpies are in agreement with tabulated thermochemical values. Thus, the linear empirical correlation between  and
and 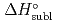 for metal-oxychlorides was updated with the inclusion of the present data. According to the predicted
for metal-oxychlorides was updated with the inclusion of the present data. According to the predicted 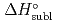 (DbOCl
(DbOCl ), a
), a  (DbOCl
(DbOCl ) value of 135
) value of 135  2 kJ/mol was extrapolated. The future accomplishment of comparative studies with DbOCl
2 kJ/mol was extrapolated. The future accomplishment of comparative studies with DbOCl under the same experimental conditions will provide valuable information on the volatility trend in Group-5 elements, together with an indication on the magnitude of relativistic effects on the electronic structure of dubnium.
under the same experimental conditions will provide valuable information on the volatility trend in Group-5 elements, together with an indication on the magnitude of relativistic effects on the electronic structure of dubnium.
 acidic solutions
acidic solutionsYokoyama, Akihiko*; Kitayama, Yuta*; Fukuda, Yoshiki*; Kikunaga, Hidetoshi*; Murakami, Masashi*; Komori, Yukiko*; Yano, Shinya*; Haba, Hiromitsu*; Tsukada, Kazuaki; Toyoshima, Atsushi*
Radiochimica Acta, 107(1), p.27 - 32, 2019/01
Times Cited Count:1 Percentile:11.15(Chemistry, Inorganic & Nuclear) glycoprotein; Microbial transformation of heavy elements in the aquatic environment
glycoprotein; Microbial transformation of heavy elements in the aquatic environmentKozai, Naofumi; Sakamoto, Fuminori; Tanaka, Kazuya; Onuki, Toshihiko; Sato, Takahiro*; Kamiya, Tomihiro*; Grambow, B.
Chemosphere, 196, p.135 - 144, 2018/04
Times Cited Count:5 Percentile:17.65(Environmental Sciences)Transformation of heavy elements by microbes such as bacteria and fungi has been an intense research subject; however, little is known about that of protozoa. This study investigated interaction of a representative protozoa, 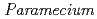 , with heavy elements (Eu(III), Pb(II), U(VI)). Non-destructive elemental analysis by micro-PIXE hardly detected those elements on living cells after sorption experiments but clearly detected on the cells that were killed with a fixative beforehand. Chromatographic analysis of aquatic species of those heavy elements after the sorption experiments revealed a fraction of those elements bound to a glycoprotein dissolved from the cell surface of living
, with heavy elements (Eu(III), Pb(II), U(VI)). Non-destructive elemental analysis by micro-PIXE hardly detected those elements on living cells after sorption experiments but clearly detected on the cells that were killed with a fixative beforehand. Chromatographic analysis of aquatic species of those heavy elements after the sorption experiments revealed a fraction of those elements bound to a glycoprotein dissolved from the cell surface of living  cells to form soluble pseudocolloid. These findings suggest that complexation of heavy elements with the dissolved surface glycoprotein reduced the sorption of those heavy elements on living cells.
cells to form soluble pseudocolloid. These findings suggest that complexation of heavy elements with the dissolved surface glycoprotein reduced the sorption of those heavy elements on living cells.
Nishio, Katsuhisa
Genshikaku Kenkyu, 62(1), p.89 - 94, 2017/09
no abstracts in English
Hofmann, S.*; Heinz, S.*; Mann, R.*; Maurer, J.*; M nzenberg, G.*; Antalic, S.*; Barth, W.*; Burkhard, K. G.*; Dahl, L.*; Eberhardt, K.*; et al.
nzenberg, G.*; Antalic, S.*; Barth, W.*; Burkhard, K. G.*; Dahl, L.*; Eberhardt, K.*; et al.
European Physical Journal A, 52(6), p.180_1 - 180_34, 2016/06
Times Cited Count:171 Percentile:93.43(Physics, Nuclear)Sato, Tetsuya
Saiensu Potaru (Internet), 3 Pages, 2015/07
On April 9th, a press release titled "Measurement of the first ionization potential of lawrencium (element 103) - Unravelling Relativistic Effects in the Heaviest Actinide Element -" was issued. This research result published from Nature was not only introduced in its "News & Views" but also appeared on the cover. I made a commentary on the result and introduced its response for public.
Oe, Kazuhiro*; Attallah, M. F.*; Asai, Masato; Goto, Naoya*; Gupta, N. S.*; Haba, Hiromitsu*; Huang, M.*; Kanaya, Jumpei*; Kaneya, Yusuke*; Kasamatsu, Yoshitaka*; et al.
Journal of Radioanalytical and Nuclear Chemistry, 303(2), p.1317 - 1320, 2015/02
Times Cited Count:10 Percentile:64.63(Chemistry, Analytical)A new technique for continuous dissolution of nuclear reaction products transported by a gas-jet system was developed for superheavy element (SHE) chemistry. In this technique, a hydrophobic membrane is utilized to separate an aqueous phase from the gas phase. With this technique, the dissolution efficiencies of short-lived radionuclides of 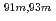 Mo and
Mo and 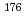 W were measured. Yields of more than 80% were observed for short-lived radionuclides at aqueous-phase flow rates of 0.1-0.4 mL/s. The gas flow-rate had no influence on the dissolution efficiency within the studied flow range of 1.0-2.0 L/min. These results show that this technique is applicable for on-line chemical studies of SHEs in the liquid phase.
W were measured. Yields of more than 80% were observed for short-lived radionuclides at aqueous-phase flow rates of 0.1-0.4 mL/s. The gas flow-rate had no influence on the dissolution efficiency within the studied flow range of 1.0-2.0 L/min. These results show that this technique is applicable for on-line chemical studies of SHEs in the liquid phase.
Komori, Yukiko*; Yokokita, Takuya*; Kasamatsu, Yoshitaka*; Haba, Hiromitsu*; Toyoshima, Atsushi; Toyomura, Keigo*; Nakamura, Kohei*; Kanaya, Jumpei*; Huang, M.*; Kudo, Yuki*; et al.
Journal of Radioanalytical and Nuclear Chemistry, 303(2), p.1385 - 1388, 2015/02
Times Cited Count:2 Percentile:17.57(Chemistry, Analytical)We studied solid-liquid extraction behavior of carrier-free Mo and W radiotracers onto an Aliquat 336-loaded resin from HCl solutions toward extraction chromatography experiments of element 106, seaborgium(Sg). Distribution coefficients (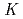
 ) of Mo and W on the resin were determined as a function of HCl concentration by a batch method. On-line extraction chromatography of Mo and W was also carried out in 2-8 M HCl solutions with an automated rapid chemistry apparatus. The order of extraction probability from 2 to 8 M HCl was Mo
) of Mo and W on the resin were determined as a function of HCl concentration by a batch method. On-line extraction chromatography of Mo and W was also carried out in 2-8 M HCl solutions with an automated rapid chemistry apparatus. The order of extraction probability from 2 to 8 M HCl was Mo W, which reflected the order of the
W, which reflected the order of the 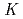
 values in the batch experiment.
values in the batch experiment.
Hayakawa, Takehito; Iwamoto, Nobuyuki; Shizuma, Toshiyuki; Kajino, Toshitaka*; Umeda, Hideyuki*; Nomoto, Kenichi*
European Physical Journal A, 27(S1), p.123 - 128, 2006/03
Times Cited Count:0 Percentile:0.01(Physics, Nuclear)no abstracts in English
Koura, Hiroyuki; Tachibana, Takahiro*
Nihon Butsuri Gakkai-Shi, 60(9), p.717 - 724, 2005/09
no abstracts in English